European registry on retinopathy of prematurity
This section keeps you updated on the major developments throughout the EU-ROP project.
If you want to receive news on the EU-ROP project by email, please register for our newsletter.
ARVO 2023
Another point on the agenda of the EU-ROP projectis of course to publish the data gathered in the EU-ROP registry.
Last month we were able to present first international data from from Germany, Turkey, Bulgaria, Austria, Ukraine, France, Poland, and Greece at the ARVO 2023 in New Orleans! It was very well taken and a lot of people got interested in the EU-ROP project.
We conducted a first analysis about the prevalence of ROP severities in our multinational cohort. The submitted abstract presents data of 203 patients from 30 centers in 8 countries with a focus on ROP characteristics such as stage, zone and presence of plus disease. We hope that by presenting the abstract at the conference it will help to raise awareness for the project and to recruit additional interested centers.
Recap 2022
In 2022, the EU-ROP project achieved many important goals. Here is a little review of the first year as an international EU-ROP registry:
After having started in August 2021, we got truly international in March 2022, when the first patient at a center outside of Germany was entered into the database.
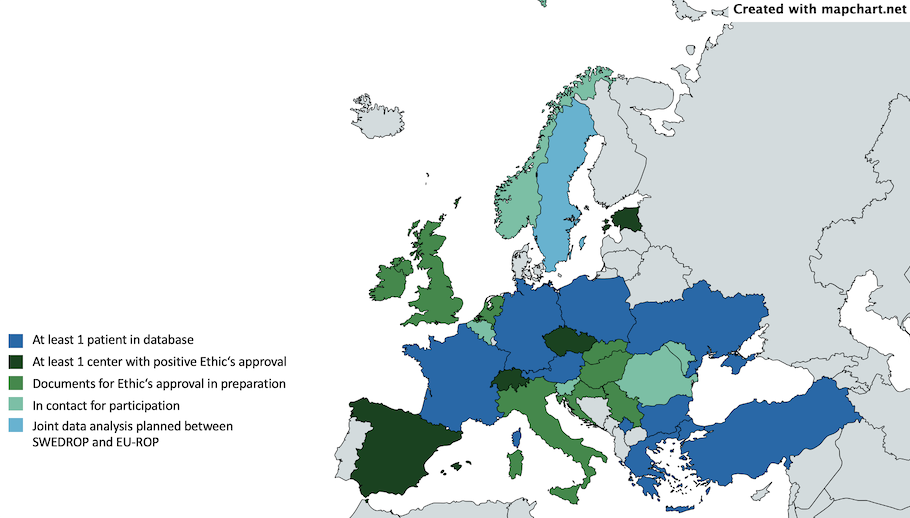
Now, only a year later there are data from 288 patients treated for ROP in the database. In just one year, eight further countries joined the EU-ROP registry and there are more to come. Three additional countries already obtained a positive Ethics’ approval and several more are currently working towards their participation.
We are very proud of how far the EU-ROP registry has come in just one year.
Of course, none of this would have been possible without the help and great work of all participating centers.
A big thank you to each and every one who is contributing to this important project!
To another year of great work and even greater accomplishments.
200th patient entered into the EU-ROP database
The EU-ROP project is growing fast. While we had celebrated the 100th patient only in June this year, we are very happy to inform you that the patient numbers have doubled by now and the 200th patient has just recently been added to the database by Dr. Ognyan Mladenov from Alexandrovska University Hospital in Sofia (Bulgaria).
This significant achievement is only possible thanks to the joint effort of all participating centers!
We have currently 31 centers from eight countries actively contributing data to the EU-ROP project. Further centers have recently obtained a positive Ethic’s vote and are about to start entering their patients.
A big Thank you to all participating centers!